
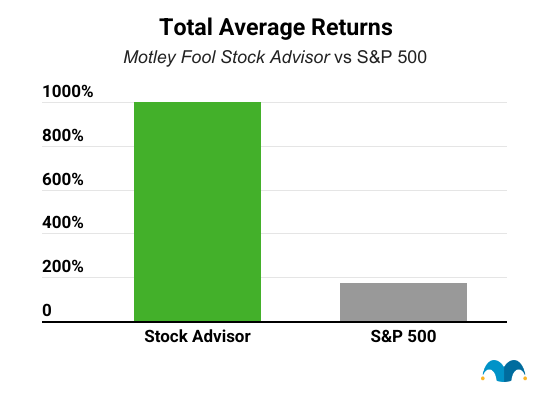
Five years from now, you'll probably wish you'd started investing with Stock Advisor
- New stock picks each month
- Detailed analysis of companies
- Model portfolios and advanced tools
- Live streaming during market hours
Featured Investing News


Want $2,000 in Annual Dividends? Invest $30,000 in These 3 Stocks

3 Top Cybersecurity Stocks to Buy in April

3 New Reasons to Like This Magnificent Dividend Stock

The Numbers Don't Lie: This 14%-Yielding Dividend Is at Risk for Another Reduction
Trending News
- Social Security's 2025 COLA Is on Track to Do Something It Hasn't Done For 15 Years
- Billionaire Bill Ackman Has His Sights on Only 1 "Magnificent Seven" Stock, and It's Not Nvidia
- The Vanguard 500 Index Fund Is Great, But Another Vanguard ETF Has Outperformed It the Past Decade
- Tesla Just Shared Some Spectacular News for Nvidia Stock Investors
- 2 Roaring Growth Stocks to Hold for the Next 20 Years
- History Says the Nasdaq Will Roar Higher This Year. My Top Growth Stock to Buy Before It Does.
- 1 Unstoppable Stock That Could Join Microsoft, Apple, Nvidia, Alphabet, Amazon, and Meta in the $1 Trillion Club
- 1 Magnificent S&P 500 Dividend Stock Down 20% to Buy and Hold Forever
- Tesla Sent a Clear Message to Wall Street on Tuesday
- This 15%-Plus-Yielding Passive Income Stock Continues to Deliver Despite Persistent Challenges
- Meet Eli Lilly's Secret Weapon in the Billion-Dollar Weight Loss Drug Market
- This Elite Dividend Stock Continues to Produce Powerful Growth
- Where Will Rivian Stock Be in 5 Years?
- 2 Fantastic Growth Stocks to Buy Right Now
- One Member of Congress Is Going Against the Grain and Selling This Skyrocketing Stock-Split Stock
Popular Topics
About The Motley Fool
Our Purpose: To make the world smarter, happier, and richer.
Founded in 1993 by brothers Tom and David Gardner, The Motley Fool helps millions of people attain financial freedom through our website, podcasts, books, newspaper column, radio show, and premium investing services. We believe that investing is empowering, enriching, and fun. We look forward to joining you on your journey to financial independence. Learn More
The Motley Fool Investing Philosophy
Learn How We Invest-
#1 Buy 25+ Companies Over Time
-
#2 Hold Stocks for 5+ Years
-
#3 Add New Savings Regularly
-
#4 Hold Through Market Volatility
-
#5 Let Your Winners Run
-
#6 Target Long-Term Returns
Investing Solutions Designed To Make Money Over The Long Term
Become a member today

Stock Advisor
Our base-level-membership giving you the foundation of a Foolish portfolio
Help members earn a positive return and build the foundations of a portfolio backed by Motley Fool principles.
Individuals who are seeking easy-to-use guidance on investing in more familiar-named companies.
$5,000 +
- Monthly stock recommendations (usually large and mid-cap stocks)
- Portfolio allocation guidance via The Motley Fool's Allocator tool
- On-going coverage of prior recommendations
- Access to Motley Fool Live video stream

Epic Bundle
A bundled membership that combines our four foundational stock-investing services into one
Help members outperform the benchmark by gaining exposure to a wider variety of stocks and investments.
Individuals who are regularly adding new money to their brokerage accounts, interested in business, and want to spend more time learning the art of investing.
$25,000 +
- Includes access to Stock Advisor + Rule Breakers + Everlasting Stocks + Real Estate Winners
- Weekly stock recommendations (ranging from small to large-cap stocks)
- Portfolio allocation guidance via The Motley Fool's Allocator tool
- On-going coverage of prior recommendations from all included services
- Access to Motley Fool Live video stream